|
And the farther they are from the righ side of the table, the weaker their electronegativity is, resulting in lower Chemical Reactivity. That is why as you go up a group Chemical Reactivity increases because it is easier for elements to gain electrons when they have high electronegativity. Chemical Reactivity  decrease as you go down  the groupįor Non-Metals, the farther right-up in the table you go, the higher the electronegativity. 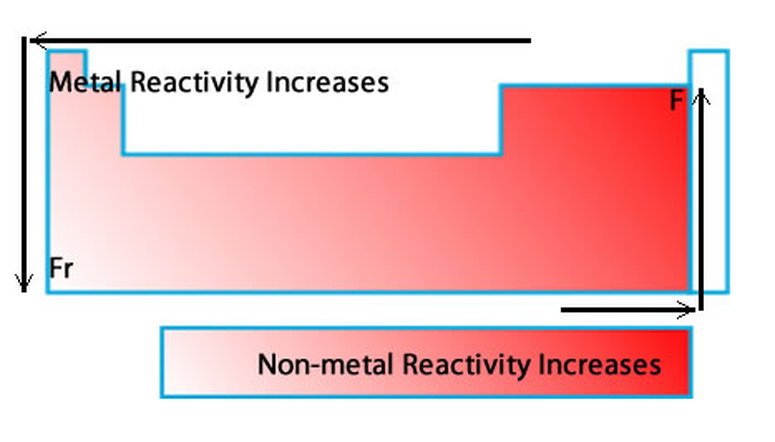 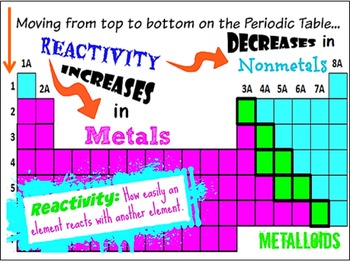 Chemical Reactivity increases as you go left to right of the periodic table.Covalent Radii of the Halogen Group Elements Atom Covalent radius (pm) Nuclear charge F 64 +9 Cl 99 +17 Br 114 +35 I 133 +53 At 148 +85 Table 6. The trends for the entire periodic table can be seen inFigure 6.31. This is due to the decreasing atomic radius and increasing nuclear charge meaning. We have quantum chemically studied the catalytic effect of various weakly interacting Lewis acids (LAs) across the periodic table, based on hydrogen (Group 1), pnictogen (Group 15), chalcogen. And you go from left to right on the periodic table elements have more electrons in their valence shells they have to get rid off which requires an element to have high energies which results into lower Chemical Reactivity. As we go across a period from left to right. First ionisation energies follow a general increasing trend along Period 2. This happens because as you go down a group, it is easier for electrons to be taken or given away, resulting in high Chemical Reactivity. Chemical Reactivity increases as you go down the group.Chemical Reactivity decrease as you go left to right of the periodic table.Īnother factor is the Electronegativity of the element, which determine how badly an element wants to take other electrons from other elements. One factor is Ionization Energy, Ionization Energy is how easily and element can remove its electrons. Chemistry - A European Journal published by Wiley-VCH GmbH.A couple factors will determine how likely and element will react to another element.  These findings again demonstrate the generality of the Pauli repulsion-lowering catalysis concept.Äiels-Alder reaction Lewis acids activation strain model density functional calculations reactivity. Notably, the reactivity can be further enhanced on going from a Period 3 to a Period 5 LA, as these species amplify the asynchronicity of the Diels-Alder reaction due to a stronger binding to the dienophile.  The reactivity of metals also increases as you go down the group. Different types of chemical bonding, and patterns and trends can be observed in their arrangement. Our detailed activation strain and Kohn-Sham molecular orbital analyses reveal that these LAs lower the Diels-Alder reaction barrier by increasing the asynchronicity of the reaction to relieve the otherwise destabilizing Pauli repulsion between the closed-shell filled Ï€-orbitals of diene and dienophile. The reactivity of metals increases from right to left across the period. The catalytic effect of various weakly interacting Lewis acids (LAs) across the periodic table, based on hydrogen (Group 1), pnictogen (Group 15), chalcogen (Group 16), and halogen (Group 17) bonds, on the Diels-Alder cycloaddition reaction between 1,3-butadiene and methyl acrylate was studied quantum chemically by using relativistic density. The periodic table arranges all chemical elements in special ways. e., the latter have the least catalytic effect. The reaction barriers systematically increase from halogen
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
